|
2/21/2023 0 Comments Electron geometry chart 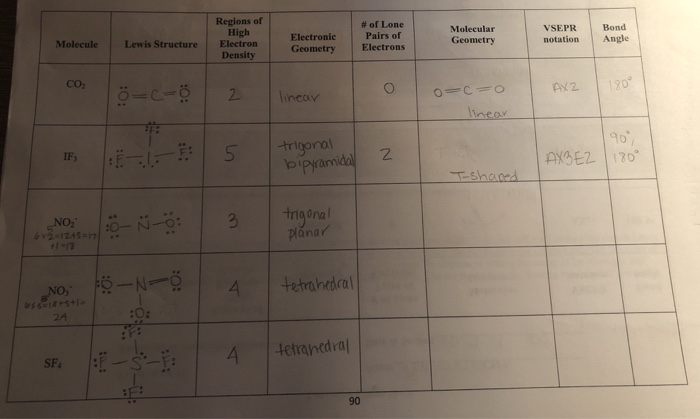
Molecular geometry, not the electron geometry (do not show lone pairs but consider them PART 3: At home: Print this table and place the 3D models on each box. 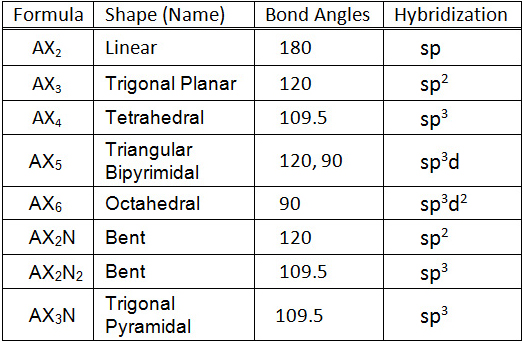
Molecule, name the geometry and write how many electrons are on the central atom. While still in the “ Real Molecules ” tab, select the following molecules. Molecule Generic Formula Generic bond angles (fromĪB 2 E 2 109 104. Select the molecules below and fill in the chart. ACE 2Ĭlick on the “ Real Molecules ” tab at the top of the page. Geometries (not electron arrangements) you don’t need to show these non-bondingĢ) Print the table in the data table section and place each molecule in theģ) Take a picture of the paper with the 3D models you made and add it to your report. When working on the 3D arrangement, but because you are representing molecular Don’t forget to consider lone pairs of electrons, The model, predict the shape the molecule will exhibit. Complete the data tables and answer the questions in the next pages.ġ) Using playdoh and toothpicks build 3D models for the following moleculesĪccording to the VSEPR theory: CO 2, H 2 O, HCN, CH 4, NH 3, SO 3.Click on reset or end the simulation and start it again to work on the activities for this.Spend a few minutes playing with the simulation, add bonds, remove bonds, add lone Go to the following website: lorado/en/simulation/molecule-shapes.To learn about how to draw 3D structures watch this video:Įxperiment courtesy of Michael Kwasny via PhET simulations/teaching materials (Original Title: 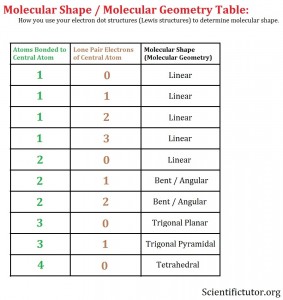
Understand more about molecular shapes watch this video: Program you are about to use allows us to visualize on a more 3-dimensional scale. It is difficult to visualize the true geometry of these molecules. Since we write in a two dimensional plane on paper, Up electrons, sometimes atoms can deviate from and not follow) the Octet Rule.īecause electrons have a negative charge and atoms occupy space, bonds and electrons But,Īs you will see, if the electronegativities are high enough and both atoms unwilling to give
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
